Featured Quizzes
User Quizzes
Create Quiz
Data and Charts
Word Search
Badges and Games
About JetPunk
Message Board
Random Quiz
Dark Mode
Radical Reactions (Organic Chemistry)
Info from Klein 4e
Rate:
Last updated: June 1, 2024
You have not attempted this quiz yet.
More quiz info >>
| First submitted | June 1, 2024 |
| Times taken | 22 |
| Average score | 66.7% | Report this quiz | Report |
4:00
The quiz is paused. You have remaining.
Copied!
Scoring
You scored / = %
This beats or equals
% of test takers
also scored 100%
The average score is
Your high score is
Your fastest time is
Keep scrolling down for answers and more stats ...
1. What picture best represents homolytic bond cleavage?
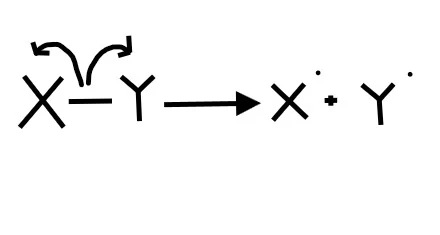
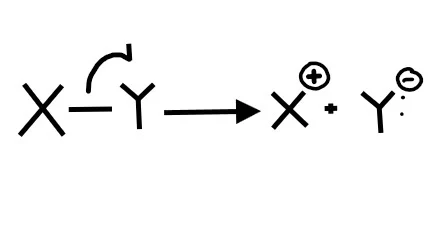
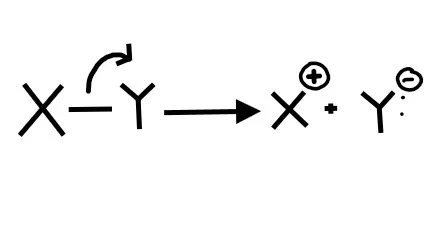
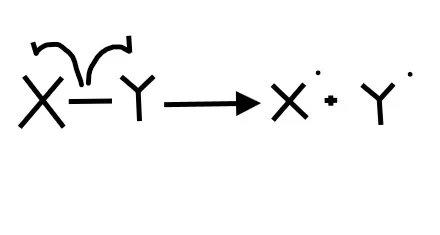
2. What picture best represents heterolytic bond cleavage?
Homolytic bond cleavage is when each element in the bond receives an electron. To show the movement of individual electrons (not an electron pair) we need to use fishook arrows

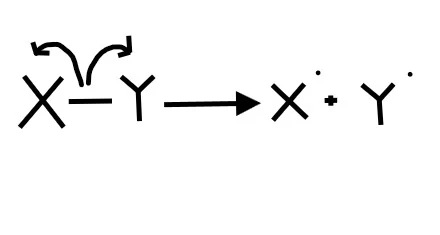
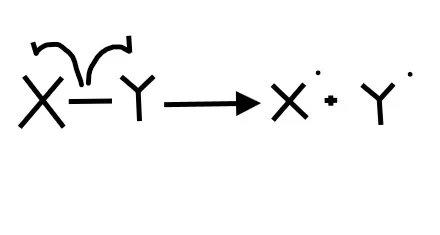

3. radicals have (effectively) a __________ geometry and are (effectively) ___ hybridized
Experimental evidence shows radicals are either trigonal planar or have a very shallow, rapidly inverting pyramid
trigonal pyramidal, sp3
trigonal planar, sp2
linear, sp
trigonal planar, sp3
4. Which radical is the most stable?
hyperconjugation
Tertiary
Secondary
Primary
Methyl
5. Which is the most stable radical?
conjugation. benzylic>allylic>tertiary
tertiary
benzylic
primary
allylic
6. Least stable radical?
methyl
secondary
primary
vinylic
7. What are the three basic stages of a radical reaction?
coupling, hydrogen abstraction, halogen abstraction
alkylation, Friedel-crafts acylation, Nucleophilic aromatic substitution
Initiation, propagation, and termination
bromination, nucleophilic attack, homolytic cleavage
8. What is a radical initiator?
light
A compound with a weak bond that uundergoes homolytic bond cleavage with ease
death
an enzyme that cleaves a radical
9. What is a radical inhibitor
alkyl peroxides
activation energy
light
A compound that prevents a chain process from getting started or continuing
Save Your Stats
Your Next Quiz
Name all 50 states in the USA. Easy, right?
Can you guess these historical and modern people who were vertically-challenged?
Find a random mystery country on the map using colour clues.
Drag the pin onto the correct country. Careful, though! Three wrong moves and the game ends.
Comments
No comments yet




